It is a known fact that Ibuprofen has been part of our daily painful life. Ibuprofen is a significant part of every athlete’s journey. It witnessed the painful struggle of almost every human being in surviving and achieving dreams; it is a critical player in the pharmaceutical industry in relieving pain. How did this wonder drug happen?
What is Ibuprofen?
Ibuprofen is frequently used, especially by athletes, to alleviate tendonitis and muscle discomfort. Ibuprofen is just one example of anti-inflammatories that can, without a doubt, greatly assist the injured athlete. However, they must be used properly and moderately. The risks of becoming dependent on these drugs much outweigh the benefits. Ibuprofen is a non-steroidal anti-inflammatory drug (NSAID). It works by reducing hormones in the body that cause pain and inflammation. Ibuprofen treats various illnesses, such as migraine, toothache, back pain, joint pain, dysmenorrhea, mild injuries, and decreased fever.
It can also be used to close a premature baby’s patent ductus arteriosus. It can be administered intravenously or orally. Usually, it starts working after an hour. Brufen, Genpril, Ibu-Tab, Cuprofen, Nurofen, Advil, and Motrin are just a few of the brand names for ibuprofen. Ibuprofen is listed among the necessary medications by the World Health Organization (WHO), a dependable source. The list outlines the essential medical requirements for a fundamental healthcare system.
Historical Timeline of Ibuprofen
Stewart Adam, a 16-year-old from Northamptonshire, invented the concept of Ibuprofen after experiencing a mild headache following a night out with his friends. He took a 600 mg dose to be safe, and it worked. Steve Adam, who dropped out of school without a clear future, had an apprenticeship in retail pharmacy at Boots, and the experience piqued his interest in a more demanding line of work. As a result, he earned a pharmacy degree from Nottingham University and a Ph.D. in pharmacology from Leeds University. In 1952, he rejoined Boots Pure Drug Company Ltd.’s research division.
His goal at the time was to discover a brand-new medication for rheumatoid arthritis that was as effective as steroids but free from their adverse side effects. He started learning about aspirin’s mechanism of action and anti-inflammatory medications because no one else seemed to be doing it. In 1897, the first non-steroidal anti-inflammatory medicine, aspirin, was created. Although aspirin was frequently prescribed as a pain reliever, it had to be administered at very high doses, increasing the risk of adverse effects such as an allergic reaction, bleeding, and dyspepsia. This indicated that by the 1950s, it was losing favor in the UK.
1953 (The Testing of Non-Hormonal Anti-Rheumatic Compounds)
The key is to create a medication that is better than aspirin, less toxic than phenylbutazone, and free of cortisone’s hormonal connections and adverse effects. It is significant to note that Adams, at this time, thought the anti-inflammatory qualities of aspirin could fully account for the analgesic action of the drug—a theory that, despite some later qualification, has shown to be at least partially true.
1956 (The Initial Experiment Stage)
This 1956 paper by Dr. Adams argued for creating a program for “non-hormonal.” At this point, Adams and one technician made up the “project” team for anti-rheumatic chemicals. Adams and Colin Burrows had already altered Wilhelmi’s UV erythema experiment in guinea pigs, which had been used to determine the anti-inflammatory effect of phenylbutazone. Trafuryl was used as the inflammogen in a later adaptation of this for the test of cutaneous erythema in humans. Later, Adams and Burrows created a more complex technique that only required a 20-second exposure to UV without anesthesia for the animal, a characteristic that not only eliminated the confusing effects of anesthesia but also allowed them to test noticeably more chemicals each day.
1961 (Filing of Patent for Ibuprofen)
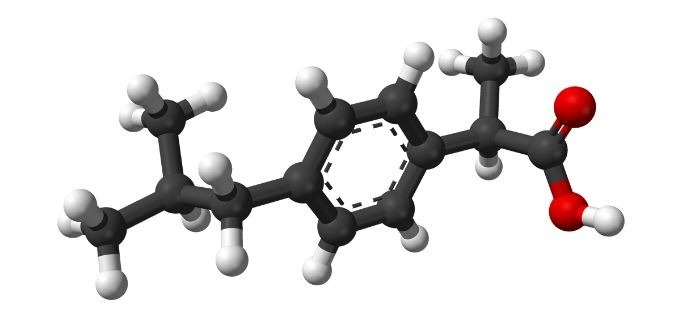
Adams and Nicholson submitted a patent application for the substance 2-(4-isobutylphenyl) propionic acid, eventually known as Ibuprofen, after vetting more than 600 candidates. The patent is awarded the following year. It’s crucial to remember that the discovery of Ibuprofen and other NSAIDs did not take place with the benefit of understanding how aspirin-like medications operated. Adams and his colleagues tested the anti-erythemic activity of several salicylates, including the hydroxylated metabolites of salicylate, that had been suggested or demonstrated to have anti-inflammatory or pain-relieving effects in rheumatoid arthritis patients.
1966 (Launching of Clinical Trial)
Numerous clinical trials have been conducted on Ibuprofen. Researchers have discovered that most Ibuprofen has high therapeutic action. It was emphasized that it is more well-tolerated than other drugs such as acetylsalicylic acid, indomethacin, and NAID, even by people with gastrointestinal lesions.
Clinical studies on individuals with rheumatoid arthritis, osteoarthritis deformans, and systemic scleroderma revealed that most patients taking Ibuprofen at daily doses of 800–1200 mg for adults and 200–600 mg for children saw a marked improvement in their overall condition, a decrease in joint pain, morning stiffness, swelling of the joints, and symptoms of carditis, as well as a lowering of the ESR and various other laboratory indicators indicative of the rheumatic process.
1969 (Launching in the UK)
Ibuprofen is sold as the prescription drug Brufen in the UK and was introduced for treating rheumatic illnesses at a dose of 600–800mg daily. Ibuprofen has a favorable side-effect profile for the gastrointestinal system compared to aspirin, the standard gold treatment for rheumatoid arthritis.
1974 (Ibuprofen was FDA Approved)
In 1974, the FDA approved Ibuprofen for sale in the US and other states. The drug has undergone several clinical studies and testing and has proven its effectiveness in treating different conditions. This medication is not only used to treat bone and muscle pain but also to treat inflammatory pain. It was sold in coated 200 mg pills, with prescriptions of 600–1200 mg per day as the starting dose. However, the daily dose can be increased to 1600 mg in emergencies and severe conditions. Moreover, the medicine should be used very carefully in patients with bronchial asthma.
1983 (Ibuprofen was Approved as an OTC medicine)
Ibuprofen was introduced as Nurofen in the UK and is approved as an over-the-counter (OTC) medication with a maximum daily dose of 1,200mg. Large-scale controlled research has backed up the relative safety of OTC ibuprofen. It has fewer gastrointestinal (GI) side effects than aspirin and the same minimal GI side effects as paracetamol.
1996 (Ibuprofen switched from OTC to GSL)
Ibuprofen has been changed to general sale list (GSL) status, allowing unsupervised sales in regular retail establishments. This medication complies with all indications, dosage, and strength requirements, and it is appropriately packaged for consumers. Consumers are informed that it is safe for public use. Ibuprofen is a medication used to treat various ailments, including headache, backache, muscle aches and pains, toothache, menstruation discomfort, and moderate arthritis pain. It also reduces the common cold-related fever.
2005 (Breast Cancer and Parkinson’s Disease Studies)
According to an observational study including 114,000 women, using Ibuprofen every day for an extended period is linked to a 51% increased risk of breast cancer, but no data regarding dosage was gathered. Moreover, according to an epidemiological study, taking Ibuprofen regularly may reduce your risk of developing Parkinson’s disease (PD). Ibuprofen may also lower the incidence of Parkinson’s disease (PD), but many concerns must be answered before it can be used, according to a 2011 Cochrane review.
2014 (More Systematic Studies were Conducted on the Effectiveness of Ibuprofen)
According to a systematic study, fast-acting ibuprofen formulations exhibit more rapid absorption, quicker pain relief, in the beginning, good overall analgesia in more individuals at the same dose, and likely longer-lasting analgesia, but no increased rate of adverse event reporting from patients. The MHRA requests the EMA to review the safety of Ibuprofen. The EMA affirms that taking large doses of Ibuprofen carries a slight risk increase for cardiovascular conditions like heart attack and stroke the following year. At OTC doses, there is no increase up to 1,200mg.
Remembering the Man Behind Ibuprofen
Ibuprofen’s inventor, who unintentionally used it to treat his hangover, passed away at age 95. The medicine underwent ten years of testing, and Dr. Stewart Adams had to wait seven years for prescription approval. After completing his pharmacy degree at the University of Nottingham, he joined Boots’ research division.
Dr. Adams’ work and contributions to patients have been praised as “inspiring” by Professor Kevin Shakesheff of the University of Nottingham. He added, “He is known for his accomplishments in developing one of the most significant analgesics ever, but like many remarkable people, he had to overcome setbacks in prior clinical studies before he and his colleagues developed Ibuprofen.
His death serves as a warning to all Nottingham residents that the work we do in our community’s businesses, hospitals, and universities has the power to alter the course of history.
The Royal Society of Chemistry awarded Dr. Adams two blue plaques for his research, and the University of Nottingham awarded him an honorary doctorate in science. He spent the remainder of his career at Boots UK, rising to the head of pharmaceutical sciences. When he spoke to the BBC in 2015, he expressed his joy at the fact that the medication he discovered is now being taken by hundreds of millions worldwide.